Climate Illuminated

CO2 Absorption Physics

Slide 10 in this series.
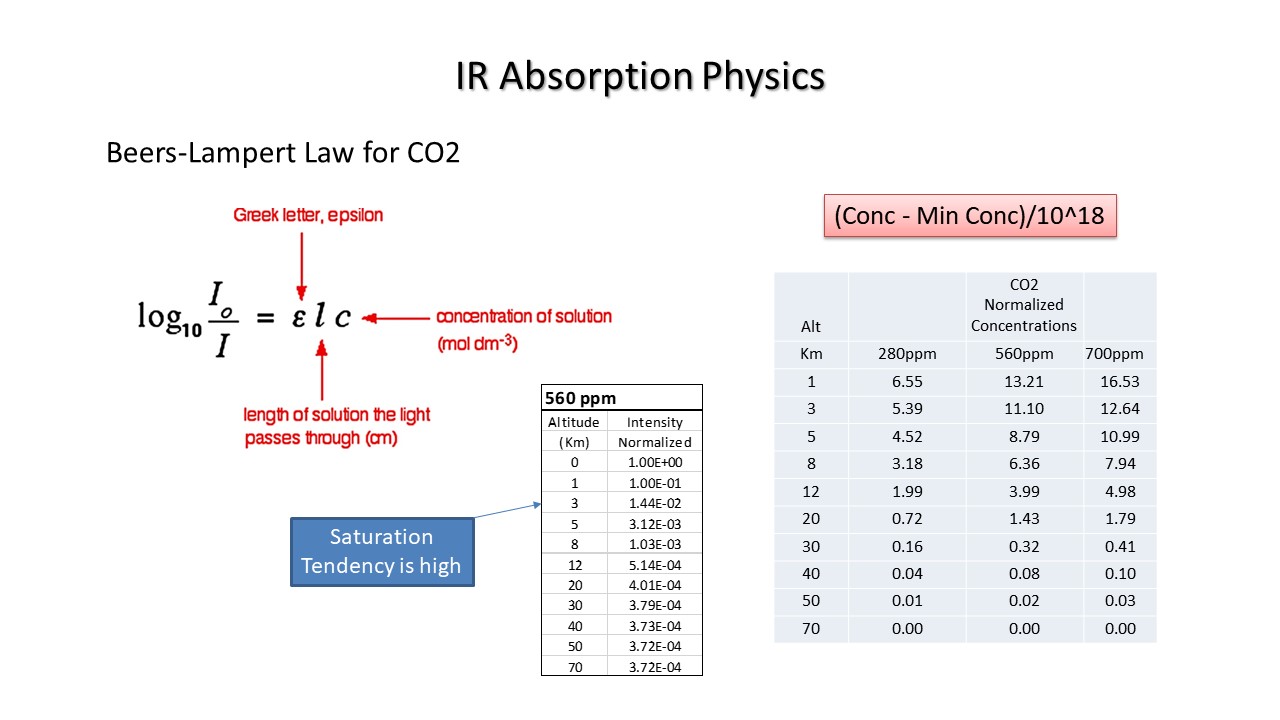
The Beers-Lampert Law is for absorption of IR in a gas media like the atmosphere. The equation is shown below and in simple terms means that the ratio intensity out to intensity in of a cube of length L is exponentially related to the concentration and the absorption. In other words the intensity falls off rapidly unless there is either a high level of the absorbing gas or a very low level.
My computations show that there is a tendency in our atmosphere to saturate at a low altitude. Some authors suggest that this saturation of absorption occurs within 1 Km of the surface.
Once absorbed what happens to the IR photon energy? It can be re-emitted or transferred into kinectic energy through collisions.
On of the serious gaps in climate science is the lack of proper theory and experiment for how collisions of various gases including water vapor with CO2 affect the transfer of this photon energy. Remember that photon energy in the long Wave lenght case is something that humans can sense as they feel heat coming from a surface.
NEXT: We can now turn to the simple AGW theory in simple equation form to better understand the underpinnings of the AGW theories.
The links below are to the pages or slides in this section, which covers the science of CO2.